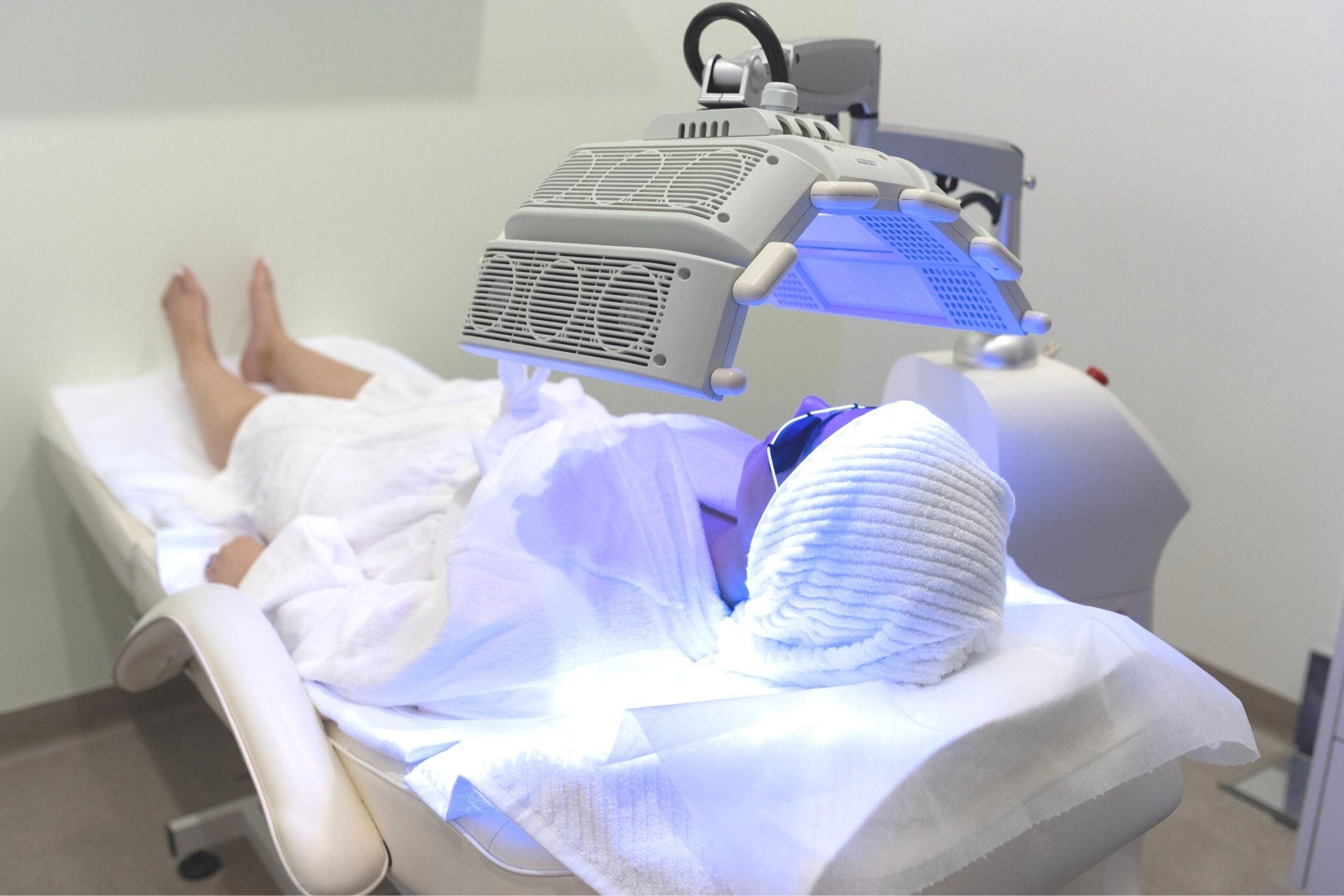
Red Light Therapy: Clinical Applications, Benefits, and Uses
Red light therapy (RLT) has gained recognition among clinics, wellness centres, and medical practices for its evidence-based applications in cellular health and recovery. For professionals asking, what is red light therapy good for, understanding both its mechanisms and clinically observed benefits is essential when integrating it into treatment offerings. Understanding